Regarding safety, the toxicity profile observed with datopotamab deruxtecan was consistent with previously reported findings. 
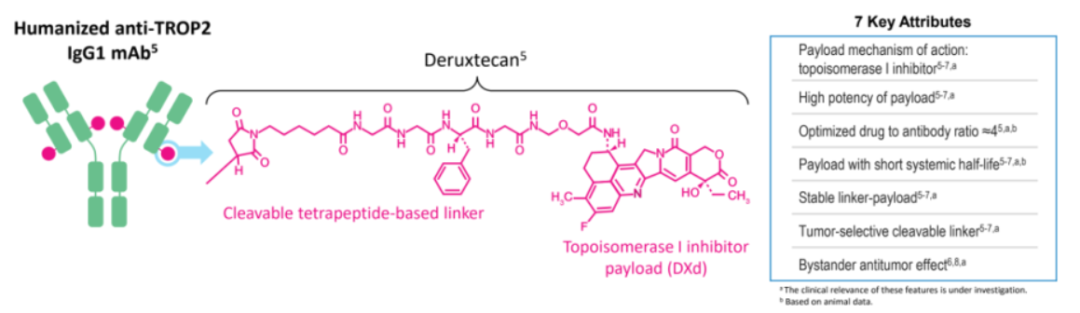
Moreover, the half-life of the agent was 4.6 days for the 6 mg/kg dose, supporting the every 3-week dosing schedule. The pharmacokinetic profile of datopotamab deruxtecan remained consistent throughout 3 cycles of treatment, irrespective of the dose. Datopotamab deruxtecan induced a disease control rate of 73% (n = 29) with 4 mg/kg, 67% (n = 26) with 6 mg/kg, and 80% (n = 64) with 8 mg/kg. Additionally, 46% of patients in the 4 mg/kg cohort, 51% of patients in the 6 mg/kg cohort, and 76% of patients in the 8 mg/kg cohort discontinued from study treatment because of progression or adverse effects (AEs).Īdditional data showed that 15% (n = 6) of patients in the 4 mg/kg group, 21% (n = 8) of patients in the 6 mg/kg group, and 9% (n = 7) of patients in the 8 mg/kg group experienced progressive disease. The median duration of follow up was 7.4 months a median of 39% of patients remained on datopotamab deruxtecan at the time of the data cutoff. The majority of patients evaluated in all 3 cohorts had received 3 or more prior lines of therapy, including platinum-based chemotherapy (median 94%), immunotherapy (median 84%), and TKIs (median 17%).A median of 37% of patients across the 3 dosing arms had a history of brain metastases and a median of 14% harbored EGFR mutations. Patients with stable, treated brain metastases were eligible for enrollment. Additionally, patients had to have an ECOG performance status of 0 or 1, and measurable disease per RECIST v1.1 criteria. Japanese patients had to be 20 years or older, and patients from the United States had to be 18 years or older to be eligible for enrollment. The ADC is comprised of a topoisomerase 1 inhibitor payload (exatecan derivative), a tetrapeptide-based linker, and a humanized Trop-2–directed monoclonal antibody.Įligible patients had to have relapsed/refractory advanced or metastatic NSCLC that was unselected for Trop-2 expression. Moreover, Trop-2 overexpression is associated with poor outcome. 
Trop-2 is a transmembrane glycoprotein that is expressed in many cancers, including lung cancer. The open-label TROPION-PanTumor01 trial marks the first-in-human study of datopotamab deruxtecan, a Trop-2–directed ADC, in patients with advanced solid tumors. The hope is not only to look at this in later lines, but also look to combine it with other agents, such as immunotherapy and potentially chemotherapy, in the future.” “Toxicity at the recommended phase 2 dose of 6 mg/kg appears to be very reasonable and is being used in the randomized phase 3 study versus docetaxel. “Datopotamab deruxtecan showed impressive response rates in a heavily pretreated population,” said Alexander Spira, MD, PhD, FACP, lead study author and director of the Virginia Cancer Specialists Research Institute and phase I trial program, in a statement to OncLive. The preliminary median progression-free survival was 4.3 months with the 4 mg/kg dose, 8.2 months with the 6 mg/kg dose, and 5.4 months with the 8 mg/kg dose.īased on these findings, the 6 mg/kg dose of datopotamab deruxtecan was identified as the recommended dose for the registrational, randomized phase 3 TROPION-Lung01 trial (NCT04656652), which is evaluating datopotamab deruxtecan versus docetaxel in patients with previously treated advanced or metastatic NSCLC who do not harbor actionable genomic alterations. Confirmed complete or partial responses were observed in 7, 6, and 19 patients, respectively.Īdditionally, 2 patients who received the 4 mg/kg dose, 2 patients who received the 6 mg/kg dose, and 1 patient who received the 8 mg/kg dose had suspected complete or partial responses, but longer follow up is needed to confirm. The preliminary efficacy results showed that datopotamab deruxtecan elicited an overall response rate of 23% (n = 9) at a dose of 4 mg/kg (n = 40), 21% (n = 8) at 6 mg/kg (n = 39), and 25% (n = 20) at 8 mg/kg (n = 80) per blinded independent central review. The investigational antibody-drug conjugate (ADC) datopotamab deruxtecan (Dato-DXd DS-1062) demonstrated encouraging antitumor activity and a manageable safety profile in patients with advanced or metastatic non–small cell lung cancer (NSCLC), according to updated results of the phase 1 TROPION-PanTumor01 trial that were presented during the International Association for the Study of Lung Cancer (IASLC) 2020 World Conference on Lung Cancer (WCLC). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
